|
Web what is the molecular geometry of pcl3 study com. View available hint (s) submitprevious answers incorrect Web in chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base. Web pocl3 molecular geometry in phosphorus oxychloride, the central atom forms bonds with four atoms. Gallery of lewis structure pcl3 : SO3 Molecular Geometry / Shape and Bond Angles (Sulfur Trioxide) YouTube Web give the electron domain and molecular geometry for p cl3 p c. 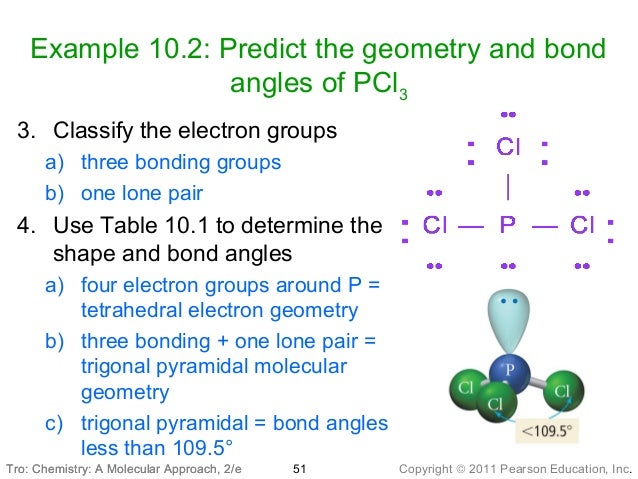 
Web using the vsepr model, what is the predicted molecular geometry of the pcl3 molecule? 60 images of lewis structure pcl3. 
Web the molecular geometry of pcl 3 is trigonal pyramidal with asymmetric charge distribution on the central atom. The lewis structure of p cl3 is the following: Web use vsepr theory to predict the shape of the pcl3 molecule. Web pcl3 molecular geometry / shape and bond angles youtube from Web use vsepr theory to predict the shape of the pcl3 molecule. Web using the vsepr model, what is the predicted molecular geometry of the pcl3 molecule? Trigonal pyramidal what is the molecular shape of the h2o molecule? Web give the electron domain and molecular geometry for p cl3 p c. Lewis dot diagram for pcl3 general wiring diagram. Enter the molecular geometry of the molecule. This is under the form of ax3e where x represents the bounded groups cl and e represents the lone. Trigonal pyramidal what is the molecular shape of the h2o molecule? What is the molecular geometry of pcl3? Web using the vsepr model, what is the predicted molecular geometry of the pcl3 molecule? Therefore this molecule is polar. Web in chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base. Phosphorus trichloride appears as a colorless or slightly yellowish liquid with a pungent odor similar to that of hydrochloric. View available hint (s) submitprevious answers incorrect Using VSEPR theory, let us try to predict the shapes of \ is trigonal pyramidal.VSEPR Valenceshell Electronpair Repulsion Theory Shows 3 VSEPR theory is based on the assumption that the molecule will take a shape such that electronic repulsion in the valence shell of that atom is minimized. The VSEPR theory is used to predict the shape of the molecules from the electron pairs that surround the central atoms of the molecule. This arrangement of the atom determines the geometry of the resulting molecule. Hint: The Valence Shell Electron Pair Repulsion Theory abbreviated as VSEPR theory is based on the premise that there is a repulsion between the pairs of valence electrons in all atoms, and the atoms will always tend to arrange themselves in a manner in which this electron pair repulsion is minimalized.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
